IS YOUR LASH GROWTH SERUM CAUSING LASTING DAMAGE? by guest blogger, Dr. Jennifer Lyerly
What do women want? The results of a 2014 Allergan survey showed 75% of women age 18-65 want longer, thicker and darker eyelashes. It was that kind of overpowering demand from market research that drove Allergan to bring Latisse to market, their blockbuster lash growth serum that shows clinically significant changes to lash length, thickness, and darkness within 16 weeks of use. The results speak for themselves:
 |
| via |
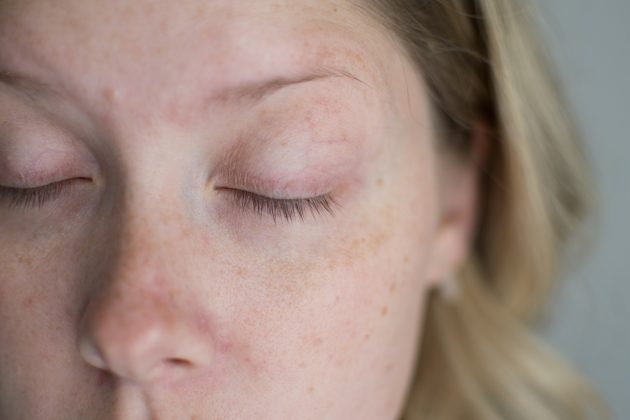 |
| Popular beauty blogger Kate of The Small Things Blog shared the dry eye side effects that made her quit Latisse on her blog. via |
It's easy to brush off eye irritation as no big deal, but ocular surface dysfunction and chronic dry eye can take a permanent and lasting toll on your vision quality and ocular comfort. Prostaglandin analogues have been used for decades in glaucoma treatment (including bimatoprost - the main active ingredient in Latisse), and studies have confirmed that they are strongly related to lasting dry eye issues. In fact just under 50% of all patients on prostaglandin analogue eye drops for the treatment of glaucoma also have a diagnosis of clinically significant chronic dry eye disease. While the preservatives in medications like Latisse or glaucoma drops have historically been blamed as the main culprit in causing dry eye, we know now that prostaglandin analogues in and of themselves can cause significant dry eye disease.
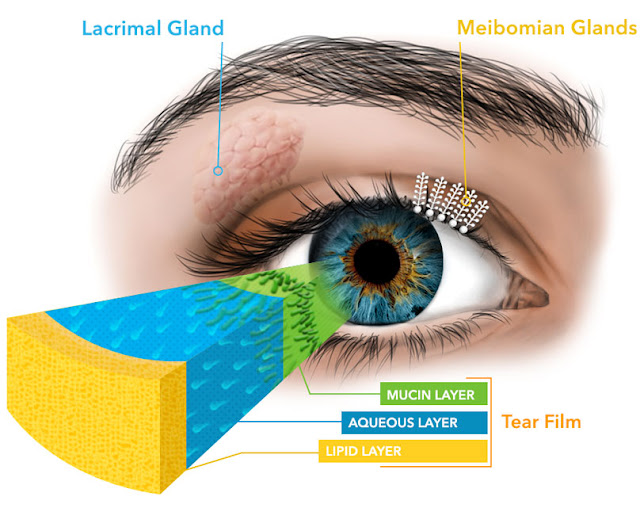
By promoting inflammation on the ocular surface, prostaglandin analogues can disrupt tear film production and expression at all layers. This includes the water layer of our tear film expressed by the lacrimal gland and the mucin layer expressed by the goblet cells on the surface of the conjunctiva. Inflammation also promotes thickening of the oil secretions (sebum) expressed by sebaceous glands in our eyelids. The meibomian (sebaceous) glands run vertically through our top and bottom eyelids and are responsible for secreting the top coat oils that hold the water and mucin layers of our tear film onto the surface of the eye.
|
| Inflammation causing dry eye can disrupt the ocular surface at every level. via |
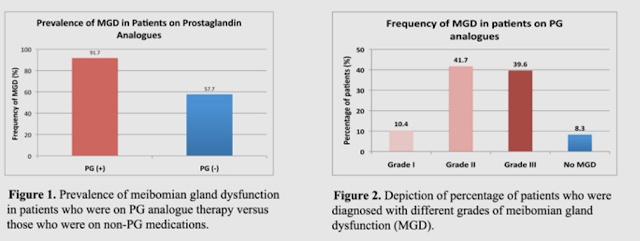
Every time we blink, the meibomian glands secrete their oil. If that oil is too thick, it's difficult for the eyelids to spread it evenly over the surface of the eye --more like toothpaste than olive oil. It may even get so thick that the oil blocks and backs up in the gland. This will slowly but surely damage the gland permanently. If the meibomian glands atrophy or die off, the body is not able to repair the tissue and the gland becomes permanently nonfunctional. The result is irreversible and often severe dry eye. A 2015 study showed a shocking 91.7% of patients treated with prostaglandin analogue drops for glaucoma had meibomian gland disease, versus only 57.7% of patients being treated for glaucoma on a different category of medication.
 |
| via |
Ingredients: Water, Butylene Glycol, Hydroxyethylcellulose, Keratin, Hydrolyzed Keratin, Biotin, Sodium Hyaluronate, Isopropyl Cloprostenate, Octapeptide-2, Allantoin, Panthenol, Copper Tripeptide-1, Pantethine, Polypeptide-23, Cucurbita Pepo (Pumpkin) Seed Extract, Glycerin, Sea Water, Malus Domestica Fruit Cell Culture Extract , Hydrolyzed Glycosaminoglycans, Prunus Amygdalus Dulcis (Sweet Almond) Fruit Extract, Backhousia Citriodora Leaf Oil, Dipotassium Glycyrrhizate, Rhizobian Gum, Styrene/Acrylates/Ammonium Methacrylate Copolymer, Xanthan Gum, PVP, Lecithin, PEG-12 Dimethicone, Alcohol Denat, Chlorphenesin, Phenoxyethanol, Sorbic Acid, Sodium Hydroxide
What is Isopropyl Cloprostenate? It's a synthetic prostaglandin analogue. As such it has the same method of action described above, and yes all the same potential side effect profile. In fact, the FDA issued a warning to OTC lash serum makers back in 2011 about the potential dangers of including a synthetic version of a prescription product with known FDA-studied side effect profile in their formulations. Statements from companies claiming that they "contain no active medical ingredient" like this from the FAQ for Rodan and Fields Lash Boost makes it all the more confusing for potential patients trying to do their due diligence if they know they have a previous diagnosis of dry eye or are at increased risk.
Here's a list of the most popular OTC lash growth serums using synthetic prostaglandin analogues in their formulas:
- Xlash Eyelash Enhancer
- Neulash
- NeuveauBrow
- RevitaLash
- Nutraluxe MD Lash
- M2 Lashes Eyelash activating serum
- Peter Thomas Roth Lashes to die for Platinum
- Rodan and Fields Lash Boost
Take Home: If you have chronic dry eye, or experience any increase in redness, watering, or eye irritation using these products, understand that you may be causing permanent damage to your delicate tear film and ocular surface. Listen to your body and discontinue use immediately if you experience any symptoms. There is a major push within the medical community to have the FDA put stronger regulations on beauty products that are using chemicals with known side effects without disclosing the risks involved, but regulation and oversight may be many years away. Just because a product is over the counter, doesn't mean that it is safe. Make sure you read the ingredients closely on all cosmetic and facial products that you use, and if you spot Isopropyl Cloprostenate in any product you are using around your eyelid, consider that lasting dry eye issues could result as a side effect.
 |
Thank you to our guest optometrist and blogger, Dr. Jennifer Lyerly. She is the founder of Eyedolatry blog, co-founder of Defocus Media and a contributing writer to the Oprah Magazine. Love Dr Lyerly? So do we! Find her and other fabulous eye-related stories at www.eyedolatryblog.com. |


